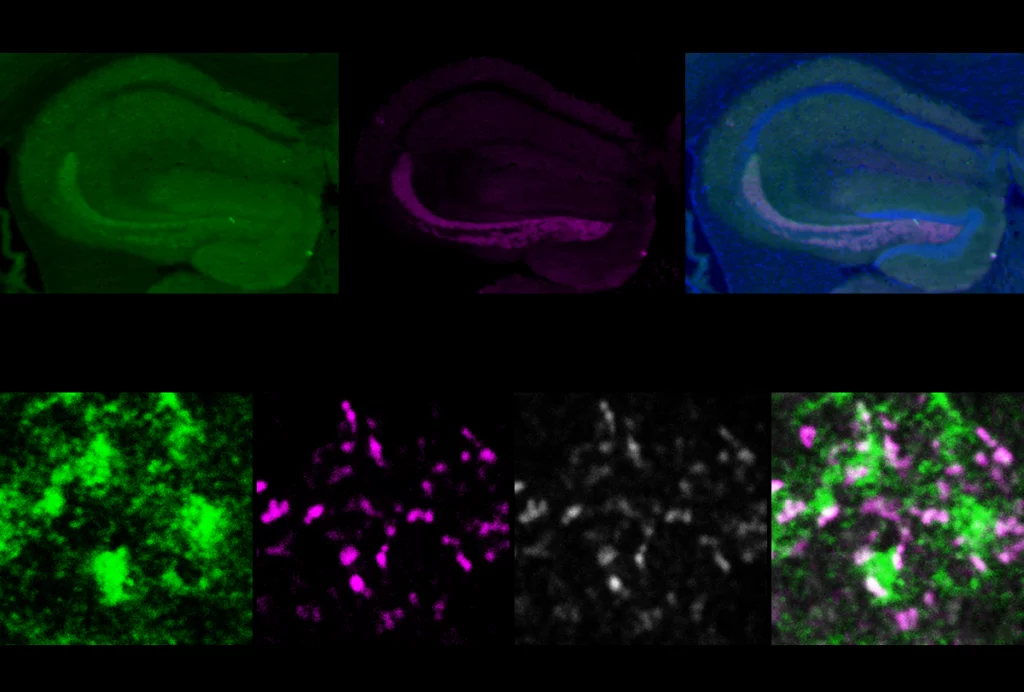
Junction function: In the mouse hippocampus, DYRK1A (green) attenuates the activity of ABLIM3 (magenta), a protein localized to tight junctions between cells (gray).
Social difficulties and deficits in inhibitory signals throughout the hippocampus are hallmarks of mice lacking one copy of the autism-related gene DYRK1A, a new study finds. Research has also shown that activating inhibitory neurons in this brain region can improve social skills in animals.
The findings reveal the neural circuits that underlie some of the difficulties associated with mutations in DRYK1A, which are associated with intellectual disability, smaller-than-average heads, seizures, and autism.
Previous studies have shown that mutations in DYRK1A affect social cognition, but the underlying circuit mechanisms were unclear, said Dr. Professor and lead researcher Amar Sahai said:
Sahay’s findings suggest that targeting the molecules and circuits downstream of the gene may reverse the social difficulties associated with DYRK1A, even in adulthood. “I don’t think we’ll ever be able to get back to the typical arc of development,” Sahay says. “Identifying opportunities for intervention in later life therefore holds great promise for new therapeutic interventions.”
This research has the benefit of approaching social cognition from a variety of angles, said Azahara Oliva, assistant professor of neurobiology and behavior at Cornell University. He was not involved in this study. Researchers are “not only narrowing down where to look in the brain, but what to look for, and even suggesting ways to repair it,” she says. [social recognition] in a medical condition. ”
TIt was reported by Sahay et al. in 2018 and 2022 that the learning process alters the inhibitory circuits in three adjacent parts of the hippocampus (CA2, CA3, and dentate gyrus). In response to experience, granule cells in the dentate gyrus increase the number of synapses with parvalbumin. neuron, a type of inhibitory neuron. These neurons increase their inhibitory contacts with excitatory neurons downstream of CA2 and CA3.
“As a result, feedforward inhibition of CA2 and CA3 cells increases depending on experience,” Sahei says. This inhibition, he says, is crucial for the simultaneous firing of neurons in groups or “ensembles” and for the generation of brain oscillations associated with learning and memory.
“Feedforward” inhibition of excitatory neurons in CA2 and CA3 also occurs when mice are exposed to stranger mice, according to a new study. This inhibition was reduced after the researchers deleted DYRK1A only in granule cells of adult mice. Unlike controls, these mice did not prefer interacting with the new mouse over the empty cup. This suggests that there are difficulties in the early stages of social cognition, often referred to as sociability. Similar circuit changes and social difficulties occurred in mice lacking systemic copies of DYRK1A.
Further experiments revealed that a protein called ABLIM3, which is expressed only in the axons of granule cells, was involved. ABLIM3 levels normally decrease in response to experience, allowing feedforward inhibition, but this was not possible in DYRK1A mice.
Attenuating ABLIM3 in granule cells of DYRK1A mice restored feedforward inhibition and alleviated the animals’ social recognition difficulties.
DYRK1A “likely downregulates or inactivates ABLIM3, resulting in increased feedforward inhibition,” Sahay says.
The researchers found that activating parvalbumin neurons in DYRK1A mice also improved the animals’ social cognition. The findings were published on October 4th. neuron.
THis findings support the hypothesis that an imbalance between inhibitory and excitatory signals in the brain may underlie some autistic traits. “People have been thinking about the balance of excitation and inhibition in autism for a long time,” says Vikas Sohal, associate professor of psychiatry and behavioral sciences at the University of California, San Francisco, who was not involved in the study. “What’s new here is that we know how [the loss of DRYK1A] In excitatory cell types, it ultimately affects the inhibition of one or two downstream synapses. ”
It is important to show that social difficulties can be reversed in adulthood, and doing so by targeting ABLIM3 or parvalbumin neurons will allow researchers to investigate the effects of loss of DYRK1A on downstream molecular and neural circuit function. Sohal says this suggests the need for a closer look.
ABLIM3 stands out among the downstream molecules of DYRK1A, and this study raises interesting questions about its role in hippocampal granule cells, said Jessica Cardin, associate professor of neuroscience at Yale University. ABLIM3 binds to actin, an essential cytoskeletal protein, and changes in the neuronal cytoskeleton are associated with autism. Cardin said this finding “suggests the possibility of structural rearrangements associated with feedforward inhibition.”
The study provides clues to potential therapeutic targets, but researchers need to investigate whether DYRK1A mutations have similar effects in different regions of the brain before moving to the clinic, she said. To tell. “There may be other pieces of the puzzle that need further consideration.”