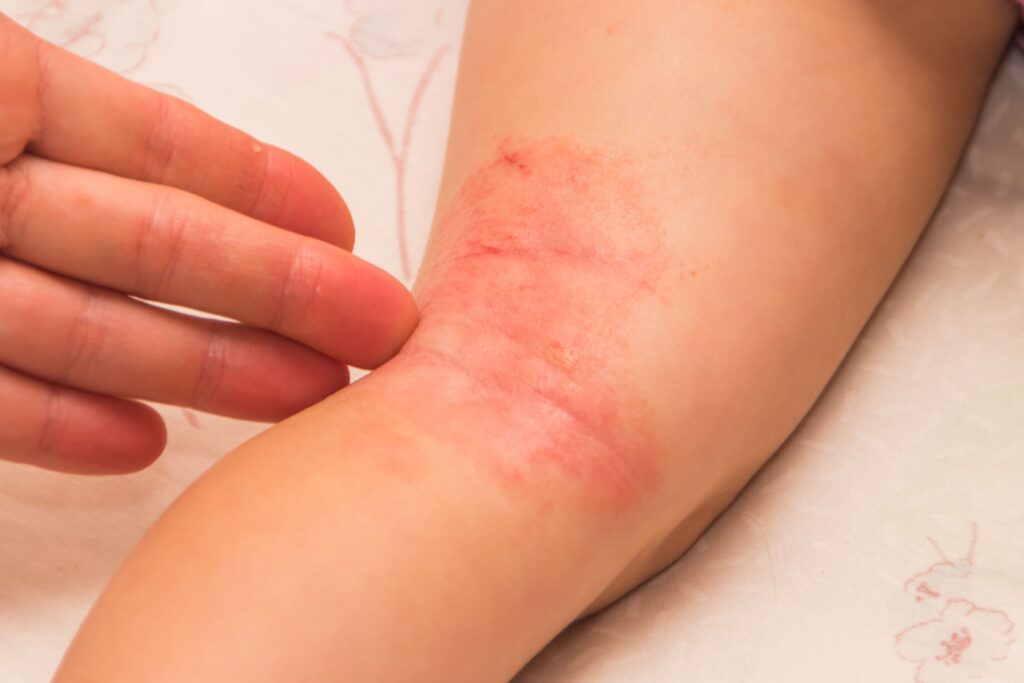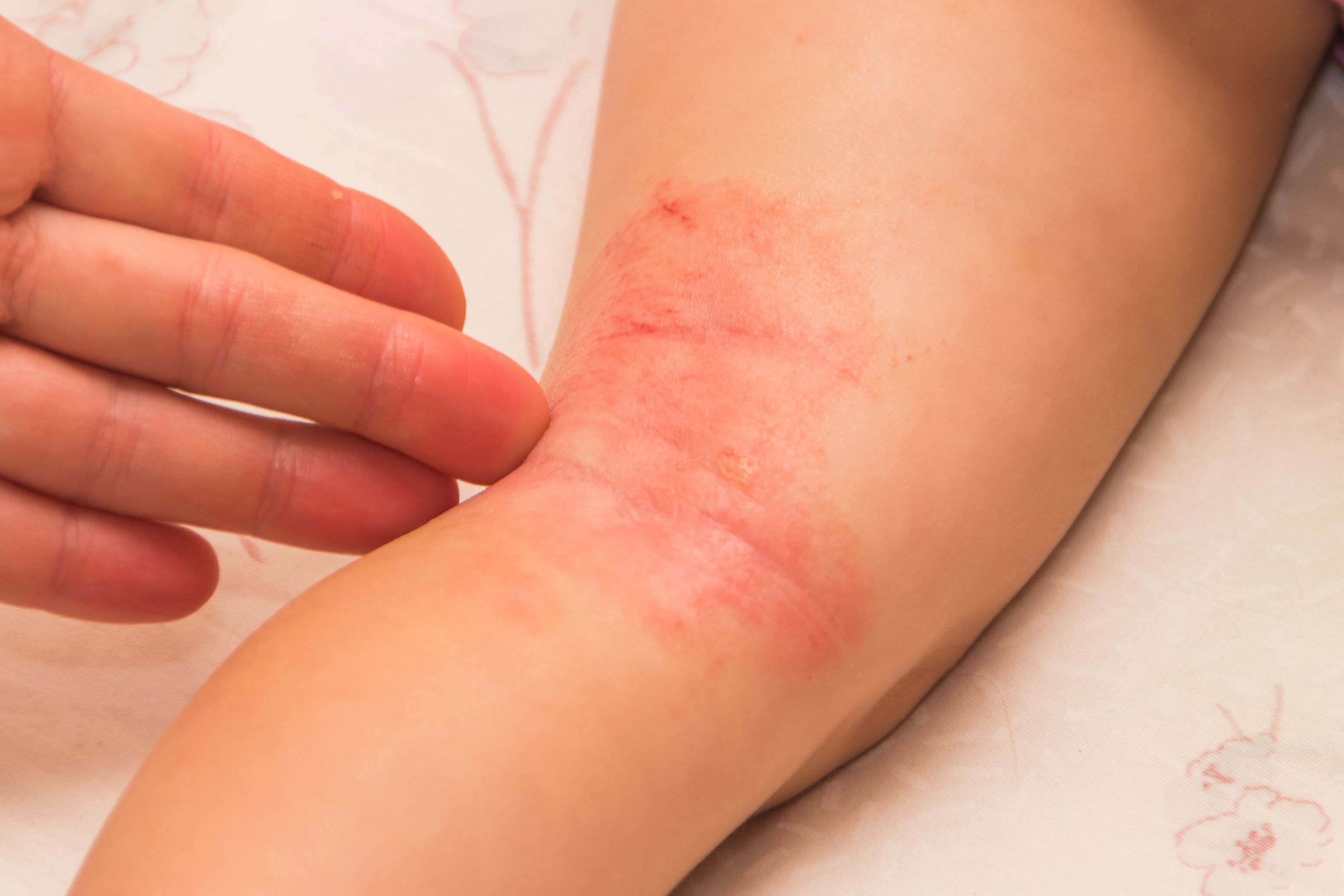
In a study of pediatric patients with atopic dermatitis (AD) treated with dupilumab, researchers found that administration of live attenuated MMR vaccine was associated with fewer serious adverse events (SAEs), regardless of the addition of live attenuated varicella vaccine. discovered that it does not cause 4 weeks immediately after vaccination.
The research was presented in a poster1 At the 2023 Academy of Dermatology Physician Assistant Fall Conference in Nashville, Tennessee.
Study authors Siegried et al. found that in pediatric patients with AD treated with dupilumab who also received live attenuated vaccination during the LIBERTY AD PRESCHOOL (NCT03346434, Part B) and LIBERTY AD PED-OLE (OLE, NCT02612454) studies. We attempted to investigate the clinical course. .
Specifically, the researchers examined nine individual patient cases of study participants with severe AD, as measured by the Investigator’s Global Rating Score of 4. These participants received live attenuated vaccination during either study, and one patient received the vaccine during LIBERTY AD. 8 of her patients vaccinated during the PRESCHOOL study and her LIBERTY AD PED-OLE.
For patients who received the live attenuated vaccine during LIBERTY AD PRESCHOOL, an interval of 12 weeks or less was added between the live attenuated vaccine and dupilumab administration. In LIBERTY AD PED-OLE, four patients in this case series had the same interval between vaccination and dupilumab administration, but four patients received vaccination and dupilumab more than 12 weeks apart.
The most common live attenuated vaccines administered to this group of patients included MMR, varicella, and DTaP vaccines.
In total, no adverse events (AEs), SAEs, or treatment-emergent infections and incursions (TEIIs) were reported among these patients within 4 weeks after live vaccine administration.
No AEs or SAEs were reported by any of the case patients in the series more than 4 weeks after vaccination. However, some TEII was reported in 4 of the 8 patients included in the case series. These include nasopharyngitis, COVID-19, molluscum contagiosum, and hand, foot, and mouth disease.
“In this limited retrospective case series of children with severe Alzheimer’s disease who also received live attenuated MMR vaccine with or without live attenuated varicella vaccine, vaccine-related No viral infections or serious adverse events were observed “4 weeks after vaccination,” Siegfried et al. wrote. “Additional studies are needed to evaluate the safety of the live attenuated vaccine in patients receiving dupilumab treatment and to investigate whether dupilumab treatment affects vaccine efficacy.”
reference
- Siegfried EC, Wine Lee L, Spergel JM, et al. Case series regarding live attenuated vaccine administration in children with atopic dermatitis treated with dupilumab. Poster presented at the 2023 Dermatology Society Physician Assistant Fall Conference held from October 26th to 29th. Nashville, Tennessee.