Key findings
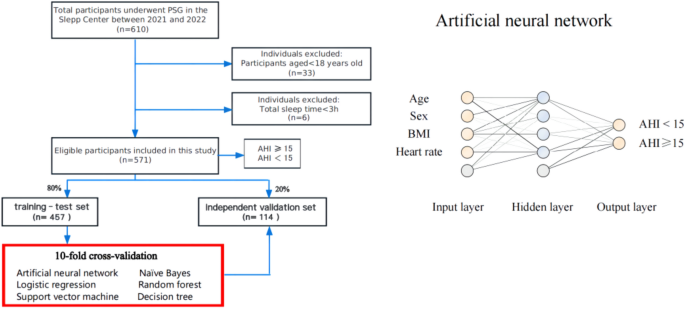
Obstructive sleep apnea (OSA) represents a significant healthcare burden worldwide, yet is largely underdiagnosed and undertreated due to low awareness and high diagnostic costs.10In this study, we developed six risk prediction models for moderate to severe OSA using four parameters: age, sex, BMI, and average heart rate during sleep. The developed models, employing LR, ANN, NB, SVM, RF, and DT, performed well in predicting moderate to severe OSA in an independent validation set, with AUROCs of 80.2%, 80.4%, 79.7%, 79.2%, 78.4%, and 70.4%, respectively. Calibration curve analysis confirmed the reliability of the models, and decision curve analysis and clinical impact curve analysis highlighted their actual clinical utility. Our models may offer the possibility of early clinical diagnosis of moderate to severe OSA in adults and reduce its adverse effects.
The model selection was mainly based on AUROC and recall metrics. Higher AUROC values indicate better predictive ability. Higher recall helps to more effectively screen patients with moderate to severe OSA, which is essential for early detection and intervention. LR, ANN, NB, SVM, RF, and DT are all widely used models in clinical medicine. ANN models that perform particularly well with high AUROC, recall, specificity, and low Brier scores likely benefit from their ability to capture nonlinear relationships in the data structure. ANN is a computational model based on the capabilities of biological neural networks that can be used to model nonlinear statistics and reveal complex relationships between input and output data, making it suitable for solving problems such as clinical prediction and image recognition.29,30.
In this study, we used a large dataset of adult hospital-based samples to make accurate comparisons between non-mild-moderate and moderate-severe OSA groups. We performed a post-hoc sample size calculation to assess whether the sample size was sufficient to draw reliable conclusions.31 This was conducted using an interactive tool available online ( https://riskcalc.org/samplesize/ ).32For the final model with four predictors, a minimum sample of 343 participants and a minimum of 22.8 events per predictor parameter were required to combine the C statistic and expected incidence to approximate the Cox–Snell R-squared.Thus, the actual sample of 571 participants in this study could provide sufficient power to ensure the reliability of the results.
Previous studies have established associations between OSA and variables such as age, sex, BMI, and average heart rate during sleep, but few studies have integrated these four factors into an OSA risk prediction model. Studies have shown that machine learning can improve the accuracy of prediction models and facilitate awareness of the underlying condition.33,34,35There are many machine learning models for predicting OSA, but most of them incorporate complex parameters that are not available in everyday life, such as cephalometric images.36Sleep Questionnaire23,36Breathing sounds during sleep37Anthropometric characteristics such as neck, waist, and head circumference23,36,38Furthermore, few predictive models have been implemented in real clinical applications.
Clinical significance
In clinical practice, our prediction model effectively classifies patients into non-mild-mild OSA and moderate-severe OSA. Patients with moderate-severe OSA exhibit more severe clinical symptoms, such as drowsiness, headache, and snoring, which have a greater impact on daily life and are more strongly associated with comorbid diseases, such as cardiovascular and cerebrovascular diseases, so it is more clinically significant to conduct early screening in these patients. The modeling parameters included in our study are simple and easy to obtain, and the generated decision curves and clinical impact curves indicate that the model has high clinical utility. In addition, the model developed in this study is based on a cloud-based mobile sleep medicine management platform for clinical application and shows unique advantages in several clinical application scenarios. For residents who undergo screening at home, it can reduce the time spent on hospital appointments and queuing, save costs, and conduct early screening in a familiar home environment, thereby increasing acceptance and cooperation. Primary hospital screening can improve the utilization rate of sleep monitoring equipment, realize medical resource and data exchange with tertiary hospitals, and provide more sleep medicine services to patients. Sleep center screening reduces the financial burden on patients, simplifies the consultation process through the use of IoT, and provides real-time initial screening results, allowing sleep physicians to advise and develop individualized treatment or referral plans. The OSA prediction model provides an effective tool for predicting or referring moderate to severe OSA under limited clinical conditions. Furthermore, the model can be applied to the general prevalence of OSA to improve people's understanding of OSA and its risks.
Limitations and future challenges
Although the model we developed shows good performance, there are some potential limitations that deserve attention and future investigation.
First, our study only used a single-center dataset that included only individuals of Asian ethnicity, therefore, we will further validate the developed model using external datasets to consider whether it can be generalized to other institutions and ethnic groups.
Second, the COVID-19 epidemic reduced the number of patients undergoing overnight polysomnography during the study period, limiting the sample size. In the future, we will conduct continuous training and optimization of the model using additional data from the sleep medicine center. Meanwhile, we will consider implementing transfer learning techniques to improve training efficiency and generalize the applicability of the model while mitigating overfitting.
Third, although participants in this study underwent PSG at a sleep medicine center and were generally considered to have a high probability of OSA or other sleep disorders, the prevalence of moderate to severe OSA was 66.4%, which is higher than the prevalence reported in the general population.6,7,39This may limit the representativeness of the model to the broader community.
Furthermore, in our retrospective study, we did not exclude individuals at high risk for undiagnosed OSA-related diseases, such as cardiovascular, metabolic, and neurological diseases. We suggest that such comorbidities may confound the model predictions and bias them towards individuals at high risk for OSA-related complications. The above indicates that our model may be suitable for detecting individuals at high risk for OSA, and the existence of collinearity problems cannot be avoided.
Fourth, the performance of our model was in the moderate range compared with previous studies, which may be related to differences in sample demographics and outcome measures. For example, the relatively young age of our study cohort suggests that the prevalence of moderate-to-severe OSA is lower compared with older populations.40,Building accurate predictive models remains a challenge.,Future efforts will focus on diversifying patient data across,different age groups to achieve a balanced dataset and ensure robust,predictive performance across diverse population segments.
Fifth, aiming for broad applicability from home environments to primary care facilities and specialized sleep centers, this model incorporates only four readily available predictors. This choice may compromise the specificity of the model in favor of simplicity. In future work, we will incorporate multidimensional indicators, such as physical examination data and laboratory indicators, to build a more accurate risk stratification model suitable for clinical practice.
Sixth, traditional questionnaire-based assessments require users to input data sequentially, which may reduce user engagement. Future research could consider AI-driven medical chatbots to increase user interactivity. Internet of Things and wearable technologies could also be applied to collect and analyze OSA-related data in real time.
Finally, as the number of people screened for OSA continues to grow, there will be increasing demands on the stability, accuracy, real-time performance, and scalability of the algorithms used. To address the computational challenges posed by large datasets, parallel computing and distributed processing techniques may need to be adopted. Furthermore, optimizing model complexity and tuning hyperparameters will be a continuous priority to align with the computational resources available at healthcare institutions.