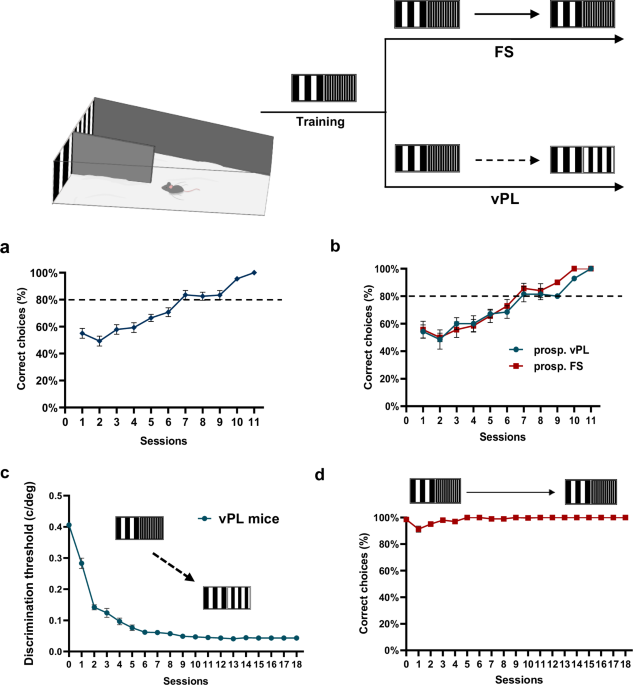
Visual perceptual learning in mice
A group of mice (n = 14) was subjected to a vPL protocol in which they were asked to discriminate two vertical gratings with equal contrast, but different spatial frequency (SF), made progressively more similar by making their SF closer to each other. Initially, the test grating had a SF of 0.522 c/deg, while the reference grating had a SF of 0.116 c/deg. All mice easily learned this discrimination task, with the percentage of correct choices increasing over the course of the training sessions (One-way RM ANOVA, Holm-Sidak method, F = 24.276, DF = 1, p < 0.001) and finally achieving a performance level of at least 80% of accuracy in at least three subsequent sessions (Fig. 1a). At this point, mice were divided in two groups: vPL mice (n = 7) underwent a perceptual learning task, practicing with a progressively more difficult discrimination as the SF of the test grating was made increasingly more similar to that of the reference grating; the other group, First-Step (FS) mice, (n = 7) continued to practice with the test grating maintained at the starting value of 0.522 c/deg. Notably, no differences were found in visual discrimination abilities between prospective FS and vPL mice during the initial phase (Two-way RM ANOVA, Holm–Sidak method, F = 1.334, DF = 1, p = 0.346, Fig. 1b).

A modified version of the visual water box task was used to perform vPL. a Mean performance in distinguishing a test grating of 0.522 c/deg from the reference grating of 0.116 c/deg across the training sessions. All animals (n = 14, vPL and FS mice) have been pooled together. The increase in the percentage of correct choices with sessions was significant (One-way RM ANOVA, Holm–Sidak method, p < 0.001). b During training, the percentage performance of prospective (prosp.) FS mice (n = 7) was not different compared to the training performance of prosp. vPL mice (n = 7, Two-way RM ANOVA, Holm–Sidak method, p = 0.346). c Improvement of discrimination threshold in vPL mice engaged in the vPL task. The threshold, calculated as the minimum spatial frequency difference between the reference and the test gratings, decreased significantly across training days (n = 7, One-way RM ANOVA on ranks p < 0.001). d After few sessions, the performance of FS mice (n = 7) engaged in a simple discrimination task remained stable across the training days. *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
In vPL mice, we observed a robust improvement in visual discrimination abilities with practice, as shown by the progressive reduction in the minimum spatial frequency discrimination threshold (MDT) across sessions. In the first session, mean MDT was 0.282 ± 0.016 c/deg, while this value reached 0.043 ± 0.004 c/deg at the end of the vPL training procedure (One-way RM ANOVA on ranks, p < 0.001, Fig. 1c). A clear vPL was also revealed by the increase in the percentage of correct choices for a given SF of the test grating (for example: reference grating of 0.116 c/deg vs test grating of 0.160 c/deg, n = 7, One-way RM ANOVA, Holm–Sidak method, F = 14.691, DF = 2, p < 0.001). In FS mice, the performance remained stable (Fig. 1d).
A separate group of animals was used to probe the difference in the vPL task between male and female mice (n = 16, 8 males and 8 females). No difference could be found either in the achieved MDT (males: 0.044 ± 0.003 c/deg, females: 0.039 ± 0.004; t test, t = 0.794, DF = 14, p = 0.441, Supplementary Fig. 1a) and the acquisition rate of the vPL (Two-way RM ANOVA, Holm–Sidak method, F = 0.521, DF = 1, p = 0.428, Supplementary Fig. 1b)
To test the specificity of vPL for the stimulus orientation, we performed, in the same vPL mice previously trained with vertical stimuli (n = 7), an experiment of orientation shift in which the two vertical gratings were rotated by 90°; afterwards, new trials were applied to assess the new MDT. We found that the visual grating discrimination abilities achieved by vPL mice were highly selective for the orientation of the stimulus; indeed, we found a marked impairment in their discrimination abilities occurring immediately after the rotation of the stimuli (n = 7, MDT before shift: 0.040 ± 0.004 c/deg, DT after shift: 0.327 ± 0.021 c/deg; paired t test, t = −13.084, DF = 6, p < 0.001, Fig. 2a, b). The animals appeared unable to discriminate the newly oriented stimuli when the test grating was maintained at the same SF reached before the orientation shift, and the percentage of correct choices fell below the 70% criterion for two consecutive vPL sessions (gray bars, Fig. 2a), until a higher SF difference between the two stimuli was achieved. With additional training, the animal performance reached a new perceptual plateau, and the MDT for horizontal grating was not significantly different from that achieved before the stimulus orientation change (n = 7, MDT before shift: 0.040 ± 0.004 c/deg, MDT after shift and vPL training: 0.040 ± 0.004 c/deg; paired t test on ranks, p = 1.000; Fig. 2a, c). In contrast, FS mice were still able to perform the task when the two vertical gratings were rotated by 90° (MDT before shift: 100% ± 0.0%; MDT after shift: 91% ± 0.035%; paired t test on ranks, p = 0.125; see Supplementary Fig. 2).

When vPL mice achieved their minimum spatial frequency discrimination threshold (MDT), the reference and test gratings were rotated by 90° keeping fixed their spatial frequency difference. a vPL mice (n = 7) were subjected to a simple incremental discrimination task until the MDT was achieved. The stimuli were then rotated by 90°. vPL mice were totally unable to discriminate the newly oriented stimuli for two consecutive sessions (performance drop below the criterion of at least 70% correct choices, gray bars). However, when additional vPL training was applied, vPL mice reached the same perceptual plateau achieved before the orientation shift. b vPL mice were totally unable to discriminate the newly oriented stimuli (n = 7, MDT before shift: 0.040 ± 0.004 c/deg, DT after shift: 0.327 ± 0.021 c/deg; two-tailed paired t test, p < 0.001). c Starting from this point, additional vPL training was applied. The new performance plateau was not significantly different from that achieved before the stimulus orientational shift (n = 7, MDT before shift: 0.040 ± 0.004 c/deg, MDT after shift and vPL training: 0.040 ± 0.004 c/deg; two-tailed paired t test, p = 1.000). *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
Electrophysiological characterization of LM activity
To provide a general characterization of basic LM neuronal activity features, visual evoked potentials (VEPs) and single-units were recorded in a group of anesthetized naïve mice (n = 7), by means of multichannel electrophysiological recordings. VEP recordings represent the elective method to study the function of visual areas and they have been extensively employed to assess visual capabilities, local cortical processing and the state of maturation of visual pathways56,57,58,59. Single-unit recordings were instead employed to locally characterize electrical responses of LM neurons.
To assess visual acuity, VEP responses were recorded from a silicon electrode inserted 3.6 mm lateral to lambda and advanced 200 μm within the cortex, in response to horizontal gratings of different SFs and maintained at 90% contrast. VEP acuity was obtained extrapolating VEP amplitude to 0 V. The average acuity was 0.408 ± 0.039 c/deg (Supplementary Fig. 3a). LM VEP acuity is just a measure of spatial resolution in LM visual neurons, but it does not necessarily reflect visual acuity in mice, as this requires recordings to be performed from V1. To measure contrast sensitivity, VEPs were recorded in response to gratings of a spatial frequency of 0.06 c/deg, at different contrasts. Contrast sensitivity was obtained extrapolating to 0 V the recorded VEP amplitudes. The average contrast threshold was 9.46% ± 2.051% (Supplementary Fig. 3b).
To further characterize the electrical activity of LM neurons, single units in response to drifting sinusoidal gratings were recorded at multiple depths, spanning all cortical layers. Single units were clustered in seven ocular dominance (OD) classes on the basis of the ratio of contralateral to ipsilateral peak responses, according to the Hubel & Wiesel’s classification60. The great majority of all recorded cells (n = 145) fell into the intermediate OD classes, without a prevalence of either the contralateral (class 1) or the ipsilateral (class 7) eye (Fig. 3a). LM electrical activity was evaluated measuring spontaneous (contra = 0.220 Hz, ipsi = 0.274 Hz, Fig. 3b) and evoked discharge (contra = 3.008 Hz, ipsi = 2.502 Hz, Fig. 3c), while orientation and direction selectivity were assessed calculating the orientation selectivity index (OSI, contra = 0.465, ipsi = 0.565, Fig. 3d) and direction selectivity index (DSI, contra = 0.225, ipsi = 0.210, Fig. 3e). No difference was found in the electrical activity recorded from the contralateral and ipsilateral eye in the spontaneous discharge or DSI (n = 6, paired t test, DF = 5, t = −1. 242 p = 0.269; t = −0.0298 p = 0.977, respectively) nor in the evoked discharge and OSI (n = 7, paired t test, DF = 6, t = 2.212 p = 0.069; t = −1.800 p = 0.122, respectively).

Single units were recorded in a group of naïve animals sampling at different cortical depths. a In order to assess LM binocularity, sorted cells were clustered in 7 ocular dominance classes according to the contralateral/ipsilateral response. Then, to characterize LM activity, the spontaneous and evoked activities (b and c, respectively), the orientation selectivity and the direction selectivity indices (d and e, respectively) were measured in all sorted units. For all the recorder parameters, no difference was found in the electrical activity measured in response to the contralateral or ipsilateral eye stimulation (n = 7, paired t test: p = 0.269, p = 0.069, p = 0.122, p = 0.122, respectively). Single dots were obtained averaging all the single units recorded in each animal. *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
Chemogenetic inhibition of LM activity
In order to silence global LM neuronal activity, a constitutive viral vector was injected into the LM of naïve mice, to induce the expression of hM4D(Gi)-mCherry, an inhibitory DREADD (designer receptors activated exclusively by designer drugs). Histological evaluations showed that the expression of hM4D(Gi)-mCherry was confined into LM, without entering the dorsal visual stream (Fig. 4).

Representation of the HVAs along the postero (P) anterior (A) and latero (L) medial (M) axes. The anatomical transition between the LM and AL cortex is considered the physiological border between the ventral and dorsal visual streams. mCherry expression was entirely confined into the ventral stream. No fluorescence could be detected within AL borders. Scale bar 500 μm; inset 200 μm. To ensure the correct localization of the injections, three random slices for each animal were aligned to Allen Mouse Brain Atlas using QuickNII. Abbreviations for visual areas: V1, primary visual cortex; A, anterior; AL, anterolateral; AM, anteromedial; LI, laterointermediate; LM, lateromedial; P, posterior; PM, posteromedial; POR, postrhinal; RL, rostrolateral.
Two weeks after the injection of the viral vector, single units were recorded from LM in a group of mice (n = 9), to probe the effective chemogenetic inhibition of LM activity. In a group of mice (n = 5), electrical signals in response to alternate monocular stimulation of the two eyes were recorded before and 15 min after an intraperitoneal (i.p.) administration of Clozapine N-oxide (CNO), a DREADD ligand. We found that CNO administration resulted in a marked suppression of the evoked activity in response to both the contralateral (pre-CNO = 2.473 ± 0.690 Hz, post-CNO = 0.789 ± 0.112 Hz; paired t test, t = 3.039, DF = 4, p = 0.038, Fig. 5a) and ipsilateral (pre-CNO = 2.559 ± 0.597 Hz, post-CNO = 0.415 ± 0.188 Hz; paired t test, t = 4.180, DF = 4, p = 0.014, Fig. 5a) eye stimulation. In contrast, no reduction in LM evoked activity was found in control mice (n = 4) recorded before (pre-SAL; contra = 1.708 ± 0.325 Hz, ipsi = 1.681 ± 0.294 Hz) and 15 min after an i.p. administration of saline (post-SAL; contra = 1.87 ± 0.358 Hz, ipsi = 1.274 ± 0.288 Hz) (paired t test, contra: on ranks, p = 0.375 and ipsi: t = 0.734, p = 0.516, Fig. 5b). Notably, LM activity recorded from CNO and SAL mice was statistically different after the i.p. administration (post CNO vs post-SAL, t test, DF = 7, contra: t = −3.242, p = 0,014 and ipsi: t = −2.733, p = 0.029). In contrast, no difference could be found between the two groups of mice before the i.p. administration of CNO or saline, respectively (pre CNO vs pre SAL, t test, DF = 7, contra: t = 1.020, p = 0.342 and ipsi: t = 1.342, p = 0.221).

Single units were recorded around 400 μm of cortical depth in a group of injected mice before and 15 min after the i.p. injection of CNO or saline, keeping all the recording parameters fixed. a A significant decrease in LM activity was detected after CNO administration (n = 5, contralateral eye: paired t test, p = 0.038; ipsilateral eye: two-tailed paired t test, p = 0.014). b In contrast, no changes in LM activity were detected after saline administration in a separated group of injected mice (n = 4, contralateral eye: paired t test, p = 0.375; ipsilateral eye: two-tailed paired t test, p = 0.516). c After CNO administration, only the LM activity recorded from post-CNO mice was significantly different from that of naïve mice (n = 6, One-way ANOVA, contralateral eye: p = 0.02 and ipsilateral eye: p = 0.031). Single dots were obtained averaging all the recorded single units in each animal. *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
To evaluate the global impact of the hM4D injection, single-unit activity recorded from CNO and SAL mice was compared to that sampled from naïve animals (n = 6). Only the evoked responses recorded from the contralateral and ipsilateral eyes of the animals treated with CNO (post-CNO) were significantly different compared to the responses recorded from naïve mice (Naïve, contra = 2.659 ± 0.354 Hz ipsi = 2.421 ± 0.603 Hz) (One-way ANOVA vs control, Holm–Sidak method, DF = 4, contra: F = 3.791, p = 0.02 and ipsi: F = 3.919, p = 0.031, Fig. 5c).
LM activity is required for vPL acquisition
Then, we used the same chemogenetic approach to test whether suppression of LM activity was able to significantly affect vPL. A group of mice (n = 16) was required to learn the vPL task two weeks after the hM4D injection. Once the 80% criterion was achieved, a subgroup of animals was subjected to the incremental phase of vPL with administration of CNO (n = 8, CNO mice), while a second subgroup of animals were tested on the same task with administration of saline (n = 8, SAL mice). Injections of either CNO or saline were performed 30 min before each vPL session. The performance plateau reached by CNO mice (0.180 ± 0.029 c/deg) was significantly different from that achieved by SAL mice (0.042 ± 0.004 c/deg), with the former group of animals displaying a robust learning impairment both in the slope of the vPL curve and in the achieved perceptual plateau (Two-way RM ANOVA on ranks, Holm–Sidak method, p < 0.001, Fig. 6a).

A group of injected mice was subjected to the vPL task 30 min after the i.p. administration of either CNO (CNO mice) or saline (SAL mice) before each session. Their vPL curves were then compared to the performance of naïve mice previously subjected to the same perceptual task (vPL mice). a CNO mice showed a strong vPL impairment during the perceptual task compared to SAL mice (n = 8 CNO and n = 8 SAL, Two-way RM ANOVA on ranks, Holm–Sidak method, p < 0.001). Compared to vPL and SAL mice, the perceptual performance of CNO mice was altered throughout the vPL task (n = 7 vPL, Two-way RM ANOVA on ranks, Holm–Sidak method, p < 0.001). CNO mice indeed displayed a different vPL progression and achieved a higher MDT. b The discrimination abilities of CNO and SAL mice were different during the incremental phase of the vPL task, but not during the training phase (n = 8 prosp. CNO and n = 8 prosp. SAL, Two-way RM ANOVA, Holm–Sidak method, p = 0.757). c To evaluate the effects of the surgical procedure on learning capability, the learning performance during the training phase of injected mice (n = 16, CNO and SAL mice pooled together) was compared to that of naïve mice (n = 14, vPL and FS mice pooled together). No difference could be found between these two groups of animals (Two-way RM ANOVA, Holm–Sidak method, p = 0.200). *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
The perceptual learning performance of CNO mice, but not that of SAL mice, was consistently different from the performance displayed by naïve animals subjected to the same vPL task (Two-way RM ANOVA on ranks, Holm-Sidak method, vPL mice vs CNO mice: p < 0.001; vPL mice vs SAL mice p = 0.513; SAL mice vs CNO mice: p < 0.001; Fig. 6a), with CNO mice reaching a significantly higher MDT compared to both SAL and vPL mice (One way ANOVA on ranks, Dunn’s method, p < 0.001). Notably, no differences could be found between mice that were administered with CNO (prospective CNO) or with saline (prospective SAL) during the learning phase of the discrimination task (Two-way RM ANOVA, Holm–Sidak method, F = 0.0980, DF = 1, p = 0.757, Fig. 6b). Moreover, the learning capabilities of the entire group of injected mice (n = 16) were not different from those of naïve mice (n = 14, vPL and FS mice pooled together), ruling out the possibility that the observed impairment in vPL animals could be dependent to learning deficits due to the surgical procedures per se (Two-way RM ANOVA, Holm–Sidak method, F = 1.694, DF = 1, p = 0.200, Fig. 6c).
To rule out the possibility that the deficits observed in CNO mice might be due to a visual impairment caused by the manipulation of LM activation, we also measured behavioral visual acuity (VA) after LM inactivation. VA was assessed through the Prusky water maze task61, testing the ability of injected mice to distinguish a visual grating from an homogeneous gray stimulus. Two weeks after a bilateral injection of hM4D, a separate group of mice (n = 8) was first trained with a low SF and then tested for the capability to distinguish higher SFs 30 min after i.p. administration of saline. At the end of this first part of the procedure, the averaged VA was of 0.514 ± 0.005 c/deg. Then, mice were retested in the same task 30 min after i.p. administration of CNO. The VA measured in the same group of mice after CNO administration was 0.516 ± 0.005 c/deg. Thus, no VA impairment could be found when LM activity was suppressed (paired t test, DF = 7, t = −0.678, p = 0.519, Fig. 7).

Visual acuity (VA) was measured in a group of injected mice to rule out any potential visual impairments caused by LM inhibition. VA was measured using the water maze test. Examples of sigmoidal extrapolations of psychometric curves used to calculate VA are reported on the top panel. a No significant differences could be found in the VAs measured before and after the chemogenetic inhibition of LM (n = 8, two-tailed paired t test, p = 0.519). b Progression in the water maze task after an i.p. administration of saline (light blue panel) or CNO (orange panel). *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
LM activity is required for vPL retention
We then asked whether the vPL impairment caused by LM inactivation could be reversed by a treatment shift in which CNO was replaced by saline administration, and vice versa, whether the intact vPL abilities displayed by mice originally treated with saline might be impaired by a treatment shift in which saline was replaced by CNO administration. To this purpose, CNO and SAL mice were subjected to an experiment of administration shift (AS). When the animals reached their perceptual plateau, CNO mice were subjected to i.p. administration of saline, and then to additional vPL practice (CNO_SAL mice); vice versa, after reaching their perceptual plateau, SAL mice were subjected to administration of CNO, and then to additional vPL practice (SAL_CNO mice) (Fig. 8). Both groups of animals were asked to perform additional vPL, starting 30 min after the first administration of the new treatment. Injections were repeated 30 min before each vPL session, as previously described for CNO and SAL mice.

a Immediately after the administration shift, no changes could be detected in the performance of CNO_SAL mice (n = 8, two-tailed paired t test, p = 0.085). After additional vPL practice, CNO_SAL mice showed a significant improvement in their perceptual performance (two-tailed paired t test, p = 0.003). Right panel: vPL progression of CNO mice before and after the administration paradigm. When LM was released from chemogenetic inhibition, the group of former CNO mice (now CNO_SAL mice) underwent a progressive vPL improvement. b Immediately after the administration shift, SAL_CNO mice could only discriminate a significantly higher DT (n = 8, two-tailed paired t test on ranks, p = 0.008). Even when additional vPL practice was applied, SAL_CNO mice were still impaired in performing the vPL task (two-tailed paired t test on ranks, p = 0.008). Right panel: vPL progression of SAL mice before and after the administration paradigm. A marked perceptual impairment was detected in the vPL performance of the former SAL mice (now SAL_CNO mice) when LM activity was suppressed, with mice being completely unable to perform the vPL task for two consecutive sessions (gray bars). *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
CNO_SAL mice displayed a marked improvement in their vPL performance. While, immediately after AS, the MDT of CNO_SAL mice was not significantly different from that achieved before AS (paired t test, DF = 7, t = 2.004, p = 0.085, Fig. 8a), they eventually reached a new MDT (0.048 ± 0.003 c/deg) that was significantly lower than that reached before AS (paired t test, DF = 7, t = 4.379, p = 0.003, Fig. 8a), and no longer different with respect to that achieved by both vPL and SAL mice (One-way ANOVA on ranks, DF = 2, p = 0.587). On the other hand, SAL_CNO mice were only able to discriminate the easiest set of stimuli of our vPL task immediately after AS (paired t test, test on ranks, p = 0.008, Fig. 8b). When additional vPL was applied, SAL_CNO mice reached a new MDT (0.319 ± 0.026 c/deg) that was significantly higher than that achieved before AS (paired t test on ranks, p = 0.008, Fig. 8b) and then that achieved by vPL mice (t test, DF = 13, t = −8.702, p < 0.001).
When LM activity was suppressed, the vPL performance of CNO, but not that of SAL mice, was significantly different compared to the performance of vPL mice- naïve animals subjected to the same vPL task (One-way ANOVA on ranks, Dunn’s Method, p < 0.001, Supplementary Fig. 4a). Instead, the MDT of SAL mice treated with CNO (SAL_CNO) but not that of CNO mice treated with SAL (CNO_SAL) was significantly different compared to the performance of vPL mice (One-way ANOVA on ranks, Dunn’s Method, p < 0.001, Supplementary Fig. 4b). Notably, we found that major perceptual impairments can be induced suppressing LM activity after vPL acquisition (SAL_CNO mice). The MDT achieved by SAL_CNO mice (MDT = 0.319 ± 0.026 c/deg) was indeed significantly higher than the MDT achieved by CNO mice, i.e. those animals subjected to LM suppression from the very first vPL session (MDT = 0.180 ± 0.029 c/deg) (t test on ranks, p = 0.021, Supplementary Fig. 4c).
Taken together, these results show that interfering with the neuronal activity of LM leads to a marked impairment not only of vPL acquisition but also of its retention (Supplementary Fig. 5a).
The role of top-down projections in vPL
To gain conclusive insights into the role played by top-down LM to V1 projections in vPL, we spatially confined our chemogenetic suppression to those secondary visual neurons directly projecting into V1 (LM > V1 projections) in a separate group of animals. To suppress LM > V1 projections, we induced the expression of a Cre-dependent hM4D delivered into LM by injecting retro-Cre within V1 borders. Through this double-injection strategy, we were able to selectively label and suppress only those LM neurons sending top-down projections to V1. As previously described for the chemogenetic suppression of LM, mice were subjected to the vPL task 30 min after an i.p. administration of CNO (PRJ CNO mice, n = 7). A significant deficit in vPL was found when LM > V1 projections where selectively suppressed, with PRJ CNO mice reaching a mean MDT of 0.235 ± 0.031 c/deg (Fig. 9a). The final perceptual plateau reached by PRJ CNO mice was not different from that previously reported for CNO mice (Two-way RM ANOVA on ranks, Holm–Sidak method, overall treatment, CNO mice vs PRJ CNO mice p = 0.333; SAL mice vs PRJ CNO p < 0.001; CNO mice vs SAL mice p < 0.001, Fig. 9a).

In a separate group of mice, we specifically targeted chemogenetic suppression to the top-down projections re-entering V1 from LM. These animals were first subjected to the vPL task 30 min after an i.p. administration of CNO (PRJ CNO mice). a) After the first session, PRJ CNO mice showed an initial vPL progression statistically different from CNO and SAL mice, and then displayed a robust vPL deficit (n = 8, Two-way RM ANOVA on ranks, Holm-Sidak method, treatment: p < 0.001). b When the perceptual plateau was achieved, PRJ CNO mice were subjected to the administration shift paradigm, as previously described for CNO and SAL mice. Immediately after saline administration, the vPL performance of PRJ CNO_SAL significantly improved (two-tailed paired t test, p = 0.084). When additional vPL practice was applied, a further improvement in the vPL performance was detected (two-tailed paired t test, p < 0.001). c When administered with CNO, PRJ CNO mice achieved an MDT different from SAL but not CNO mice (n = 8 per group, One-way ANOVA on ranks, p < 0.001), displaying a clear vPL impairment. d In contrast, PRJ CNO_SAL achieved an MDT different from SAL_CNO but not CNO_SAL mice (n = 8 per group, One-way ANOVA on ranks, p < 0.001). Immunohistochemistry labeling: red-mCherry, green-eGFP, yellow-merge, blue-Hoechst. *Statistical significance. Error bars, s.e.m. Source data are provided as a Source Data file.
We then tested whether the vPL deficit exhibited by PRJ mice could be rescued by a shift to saline administration, as previously observed in CNO mice. When PRJ CNO mice achieved their perceptual plateau, they were subjected to additional vPL practice 30 min after a saline i.p. administration (PRJ CNO_SAL mice). When LM > V1 projections were released from the chemogenetic suppression, PRJ CNO_SAL mice showed an improvement in their performance (0.235 ± 0.031 c/deg vs 0.188 ± 0.039 c/deg; paired t test, t = 2.576, DF = 6, p = 0.042, Fig. 9b), continuing to improve until they achieved a new MDT (0.043 ± 0.006 c/deg, paired t test, t = 6.606, DF = 6, p < 0.001, Fig. 9b).
Similarly to what was observed when the global LM activity was suppressed, we found that PRJ CNO mice achieved a vPL plateau statistically different from SAL mice, but not from CNO mice (One-way ANOVA on ranks, p < 0.001, Fig. 9c). However, when the activity of LM > V1 projections was rescued, PRJ CNO_SAL mice achieved a new perceptual plateau statistically different from SAL_CNO mice, but not from CNO_SAL mice (One-way ANOVA on ranks, p < 0.001, Fig. 9d).
Overall, these results strongly show a key role for top-down projections that re-enter V1 from LM in the acquisition of vPL (Supplementary Fig. 5b).